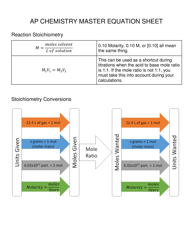
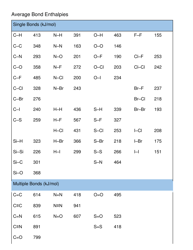
Your Complete Guide To The AP Chem Equation Sheet
Are you feeling overwhelmed by the sheer volume of equations, constants, and formulas you need to know for the AP Chemistry exam? You're not alone. Every year, thousands of students stress about memorizing every single formula, but what if we told you there's a better way? The AP Chemistry Equation Sheet is your secret weapon for exam success, and understanding how to use it effectively can make all the difference in your performance.
Understanding the AP Chemistry Equation Sheet
The AP® Chemistry Equations and Constants document is a comprehensive reference sheet that will be provided to you during both sections of the AP Chemistry exam. This invaluable resource contains all the essential equations, constants, and unit conversions you'll need to solve problems efficiently. Let's dive into what exactly this sheet includes and how you can make the most of it.
Key Units and Symbols
The equation sheet includes a standardized set of unit symbols that you'll encounter throughout the exam. These fundamental units form the building blocks of all chemical calculations:
- Jasmine Crockett Dating
- 5movie Rulesus
- You Wont Believe This Leak About Robbie Pardlo Its Absolutely Devastating
- Liter (L) - volume measurement
- Meter (m) - length measurement
- Second (s) - time measurement
- Hertz (Hz) - frequency measurement
- Atmosphere (atm) - pressure measurement
- Millimeter of mercury (mmHg) - pressure measurement
- Degree Celsius (°C) - temperature measurement
- Kelvin (K) - absolute temperature measurement
- Joule (J) - energy measurement
- Volt (V) - electrical potential measurement
- Coulomb (C) - electric charge measurement
- Ampere (A) - electric current measurement
Understanding these units and their relationships is crucial for solving problems efficiently during the exam.
What's New for 2025
AP® Chemistry Equations and Constants, effective 2025 brings some important updates that students should be aware of. The College Board has refined the equation sheet to make it more user-friendly and comprehensive. The latest version includes:
- Unit conversions clearly organized for quick reference
- Updated constants that reflect the most current scientific data
- Streamlined organization to help you find information faster
- Additional formulas that align with the latest curriculum framework
This updated sheet is designed to reduce memorization burden while ensuring you have access to all necessary information during the exam.
- Emotional Explosion What Your Heart Truly Wishes For In Every Dream
- Sinbad Legend Of The Seven Seas A Dreamworks Animated Classic
- Sophie Rain Profession
Downloading Your Study Resource
You can download a PDF file with the most common symbols, formulas, and constants for AP Chemistry exams from the College Board website. Having this document readily available for your study sessions is essential. Here's why:
Familiarity: The more you work with the official equation sheet during practice, the more comfortable you'll be using it during the actual exam.
Efficiency: Knowing exactly where to find specific equations saves precious time during the test.
Confidence: Understanding the layout and content of the sheet reduces anxiety and improves performance.
Comprehensive Topic Coverage
The equation sheet covers all major topics you'll encounter on the AP Chemistry exam, including:
Atomic Structure
- Electron configuration
- Quantum numbers
- Atomic spectra
- Periodic trends
Equilibrium
- Equilibrium constants (K and Q)
- Le Chatelier's principle applications
- Solubility product constants
- Acid-base equilibrium expressions
Kinetics
- Rate laws
- Integrated rate equations
- Arrhenius equation
- Reaction mechanisms
Gases
- Ideal gas law
- Gas law variations
- Kinetic molecular theory
- Gas stoichiometry
Liquids and Solutions
- Concentration units
- Colligative properties
- Raoult's law
- Henry's law
Thermochemistry
- Enthalpy changes
- Hess's law
- Calorimetry equations
- Gibbs free energy
Electrochemistry
- Cell potentials
- Nernst equation
- Faraday's laws
- Electrolysis calculations
The Good News for AP Chemistry Students
Good news for AP Chemistry students: You don't have to memorize every single equation. The equation sheet is provided for both sections of the exam, meaning you can focus your study efforts on understanding concepts rather than rote memorization of formulas.
This approach allows you to:
- Focus on application: Understanding when and how to use equations is more important than memorizing them.
- Reduce stress: Knowing you have a reliable reference reduces exam anxiety.
- Improve problem-solving: You can spend more time analyzing problems and less time trying to recall formulas.
Best Practices for Using the Equation Sheet
While having the equation sheet is incredibly helpful, there are some important strategies to maximize its effectiveness:
Always Balance Equations First
Always balance equations first - this fundamental rule cannot be stressed enough. Before applying any formulas from the equation sheet, ensure your chemical equations are properly balanced. This step is crucial because:
- It ensures the correct stoichiometric relationships
- It prevents calculation errors
- It helps you identify the appropriate formulas to use
Understanding vs. Memorization
The equation sheet is a tool, but it's not a substitute for understanding. You should know:
- What each equation means: Understanding the physical significance of equations helps you apply them correctly.
- When to use each equation: Different situations call for different formulas.
- How to manipulate equations: You may need to rearrange formulas to solve for different variables.
Practice with the Sheet
Need help mastering these formulas? The best approach is consistent practice with the official equation sheet. Work through practice problems while referring to the sheet, gradually building your familiarity and speed.
Expert Support for AP Chemistry Success
Our Ivy League chemistry tutors are here to help you reach your target score. Professional tutoring can provide:
- Personalized guidance: Targeted help on your specific areas of difficulty
- Test-taking strategies: Expert techniques for efficient problem-solving
- Confidence building: Support that helps you approach the exam with assurance
If you're struggling with certain concepts or want to maximize your score potential, consider seeking expert assistance.
Getting Started with Professional Help
Book a free consultation on our site to discuss your AP Chemistry needs. During this consultation, you can:
- Discuss your current performance and goals
- Identify areas where you need the most support
- Learn about tutoring options and packages
- Get answers to your specific questions
Professional guidance can make a significant difference in your understanding and performance.
Study Strategies for Maximum Effectiveness
To make the most of your equation sheet and overall AP Chemistry preparation:
Create a Study Schedule
Develop a consistent study routine that includes:
- Regular review of key concepts
- Practice problems using the equation sheet
- Time management practice for exam conditions
- Periodic self-assessment to track progress
Use Active Learning Techniques
Engage with the material through:
- Flashcards for key concepts and equations
- Practice tests under timed conditions
- Study groups for collaborative learning
- Teaching others to reinforce your own understanding
Focus on Weak Areas
Identify topics where you struggle and dedicate extra time to:
- Reviewing fundamental concepts
- Working additional practice problems
- Seeking help when needed
- Building confidence through mastery
The Ultimate Study Aid
With clear, concise explanations and organized sections, this cheatsheet is an invaluable study aid for achieving a high score on the AP Chemistry exam. The equation sheet serves as more than just a reference during the test—it's a comprehensive study tool that can guide your preparation throughout the year.
Atomic Structure & Properties: The Atom
Understanding atomic structure forms the foundation for much of AP Chemistry. The equation sheet includes essential information about:
- Subatomic particles: Protons, neutrons, electrons
- Atomic models: Historical development and modern understanding
- Quantum mechanics: Wave functions and probability distributions
- Periodic trends: Atomic radius, ionization energy, electron affinity
Mastering these concepts is crucial because they underpin many other topics in chemistry.
Conclusion
The AP Chemistry Equation Sheet is truly your secret weapon for exam success. By understanding its contents, practicing with it regularly, and focusing on conceptual understanding rather than memorization, you can approach the AP Chemistry exam with confidence.
Remember these key takeaways:
- The equation sheet is provided for both sections of the exam
- Understanding when and how to use equations is more important than memorizing them
- Regular practice with the sheet builds familiarity and efficiency
- Professional help is available if you need additional support
- Conceptual understanding combined with strategic use of the equation sheet leads to success
Your journey to AP Chemistry success starts with understanding and effectively utilizing the resources available to you. The equation sheet is more than just a reference—it's a roadmap to mastering the material and achieving your target score. With proper preparation and the right strategies, you can turn what seems like an overwhelming amount of information into manageable, masterable concepts.
Good luck with your AP Chemistry preparation!